Why weight sorting?

What are precision weight sorters and checkweighers ?
Precision weight sorters and checkweighers are highly accurate devices which can rapidly check large quantities of product, separating out items that are within acceptable limits from those that are under or over-weight. These devices weigh each tablet or capsule individually and can sort an entire batch unattended while keeping detailed records of every weighing.Who uses precision weight sorters and checkweighers?
Within the healthcare sector, weight sorters and checkweighers are widely used by lab staff in research and development facilities, including the people who are preparing and verifying doses for clinical trials. They are also used by people who are responsible for manufacturing pharmaceutical, nutraceutical and veterinary medicine.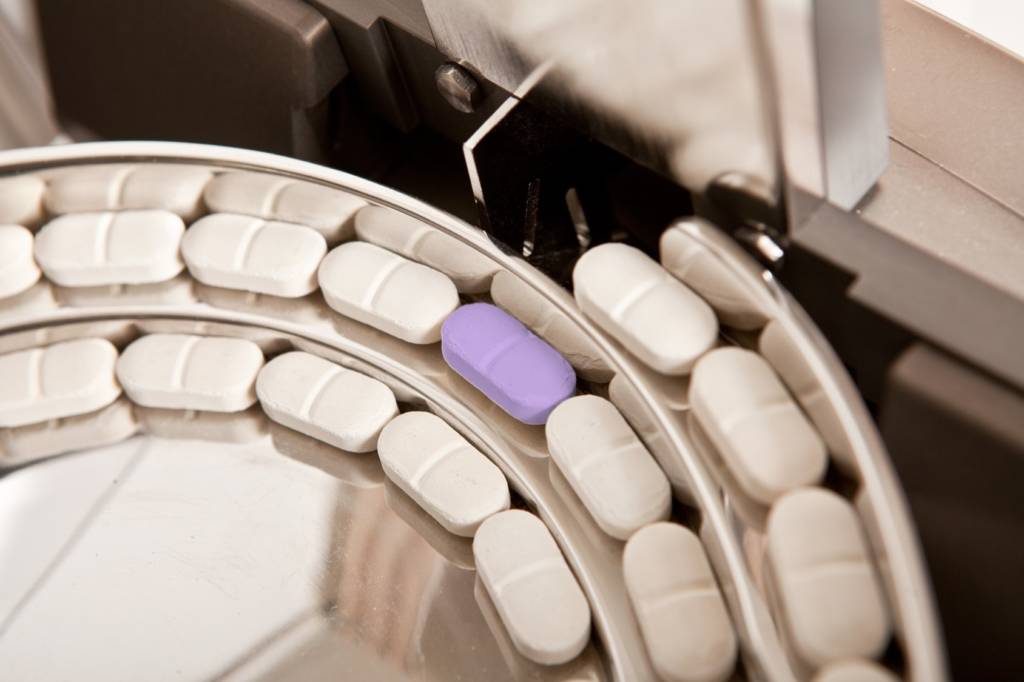

What are the applications for weight sorting?
In research and development, product for clinical trials is 100% weight checked for compliance. Traditionally this is slow, laborious and error-prone; it ties-up skilled technicians who could be doing more valuable work. In contrast, with a weight sorter every item is checked automatically in a rapid, reliable and unattended process. In healthcare manufacturing, all organisations still experience some batches which are rejected due to weight issues or damaged product. These represent a large cost, in terms of wasted ingredients, production time and disposal. Using a precision weight sorter, an entire batch can be checked so that all the defective items are separated out and typically over 90% good product recovered.How good is the return on investment?
Precision weight sorters and checkweighers offer outstanding return on investment. A machine purchased for batch recovery will typically pay for itself in its first use. The business case for weight sorters in research and development is equally compelling, with labour savings readily covering the investment, alongside reduced compliance risks.
Find out more
To find out more, please read our topics, recovering rejected product and cost-effective compliance in R&D or contact us.Explore More
Continue Your Research
Individual OSD weight sorting is a vital process for quality control
Pharmaceutical R&D, clinical trials and manufacture all require strict standards of uniformity, and weight is..
Contact UsExplore our range of industry-leading weight sorters
As leaders in precision weight sorting, we help businesses achieve the highest levels of quality assurance with our weight sorters through unrivalled accuracy, efficiency and reliability.
More5 Factors to Consider When Choosing Your MES Partner
Selecting the right partner is as important as choosing the right MES solution. We understand..
Contact UsHow you can meet us
Request a meeting, or find us at an exhibition
Request a meetingUpcoming exhibitions




