Ci-DMS MES – Manufacturing Execution System
Upgrade your weigh & dispense operation to gain the benefits of a full MES solution
The Ci-DMS MES is a manufacturing execution system designed specifically for the healthcare industry, giving you the benefits of electronic control of your production processes and built-in compliance.
Developed according to GAMP standards, the Ci-DMS MES allows you to electronically manage all stages of batch manufacture from weigh and dispense to packing for a wide range of final dosage forms. The system is designed to help you meet your data integrity obligations with particular emphasis on data security, electronic signatures and audit trails.
- Runs on fixed terminals or mobile wireless devices for full flexibility in managing production workflows
- Replaces paper-based processes while improving efficiency, productivity and compliance
- Generates comprehensive reporting and management information from electronically captured data
- Helps you achieve standard manufacturing processes with electronic SOPs
- Eliminates the requirement for a manual check of every operator action
- Phased implementation delivers faster ROI
Manufacturing
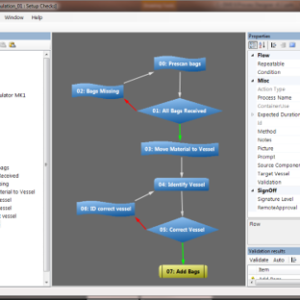
The Ci-DMS MES electronically controls the actions required for capturing batch manufacturing information.- All paper-based processes can be managed electronically
- All batch manufacturing processes can be designed electronically (including recipes, work instructions, cleaning, equipment usage and corrective actions)
- Interfaces available for a range of manufacturing hardware
- Every batch action is captured electronically and can be configured to require an electronic signature
- Typical ROI of less than two years
Ci-DMS MES in the Granulation Suite
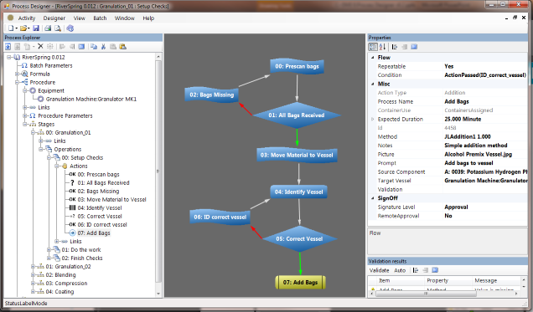

Manufacturing Operations
Ci-DMS provides you with the tools you need to control the day-to-day running of your batch manufacturing operations from batch scheduling to the generation of GMP batch reports and analysis of manufacturing efficiency. Powerful GMP, in-process and cross batch analysis information helps reduce batch release times and improve production efficiency. Browser-based reports can be viewed securely from any web-enabled location.- Manages master data, hardware and system security
- Flexible scheduling capability for products and ingredients
- Comprehensive electronic signature and audit trail features
- Highlights batch production issues in real time
- Enables faster batch review and release
- Cross batch analysis capability helps reveal trends and inefficiencies




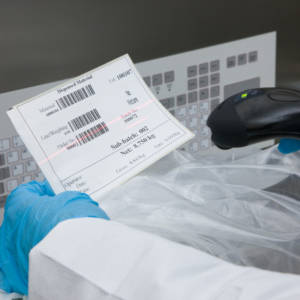
Weigh and Dispense
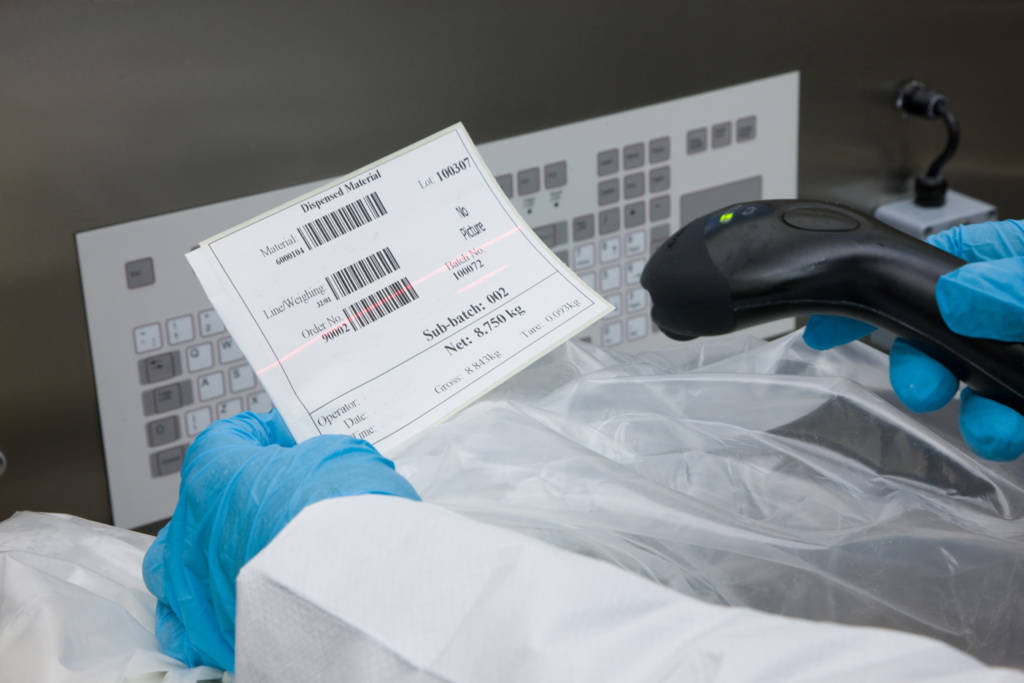
The Ci-DMS manufacturing execution system electronically manages the weigh and dispense requirement for manufacturing and packing materials and raw material sampling.- Fully configurable interfaces with scales, barcode scanners and label printers
- Faster material staging and more efficient booth utilisation with campaign weighing
- Replaces all paper-based operations including booth cleaning logs, balance validation logs, container labelling and batch manufacturing records
- All significant actions can be configured to require an electronic signature
- Can provide real-time stock consumption updates to your host system
- Typical ROI of less than one year
Ci-DMS Weigh & Dispense in action



Packing
Ci-DMS electronically controls the actions required for electronically recording batch packing information.- All paper-based processes can be managed electronically
- All batch packing processes can be designed electronically (including recipes, work instructions, equipment usage and sampling rules)
- Supports cleaning, equipment validation and corrective action processes
- Every batch action is captured electronically and can be configured to require an electronic signature
- Typical ROI of less than two years
Ci-DMS MES in the Packing Area
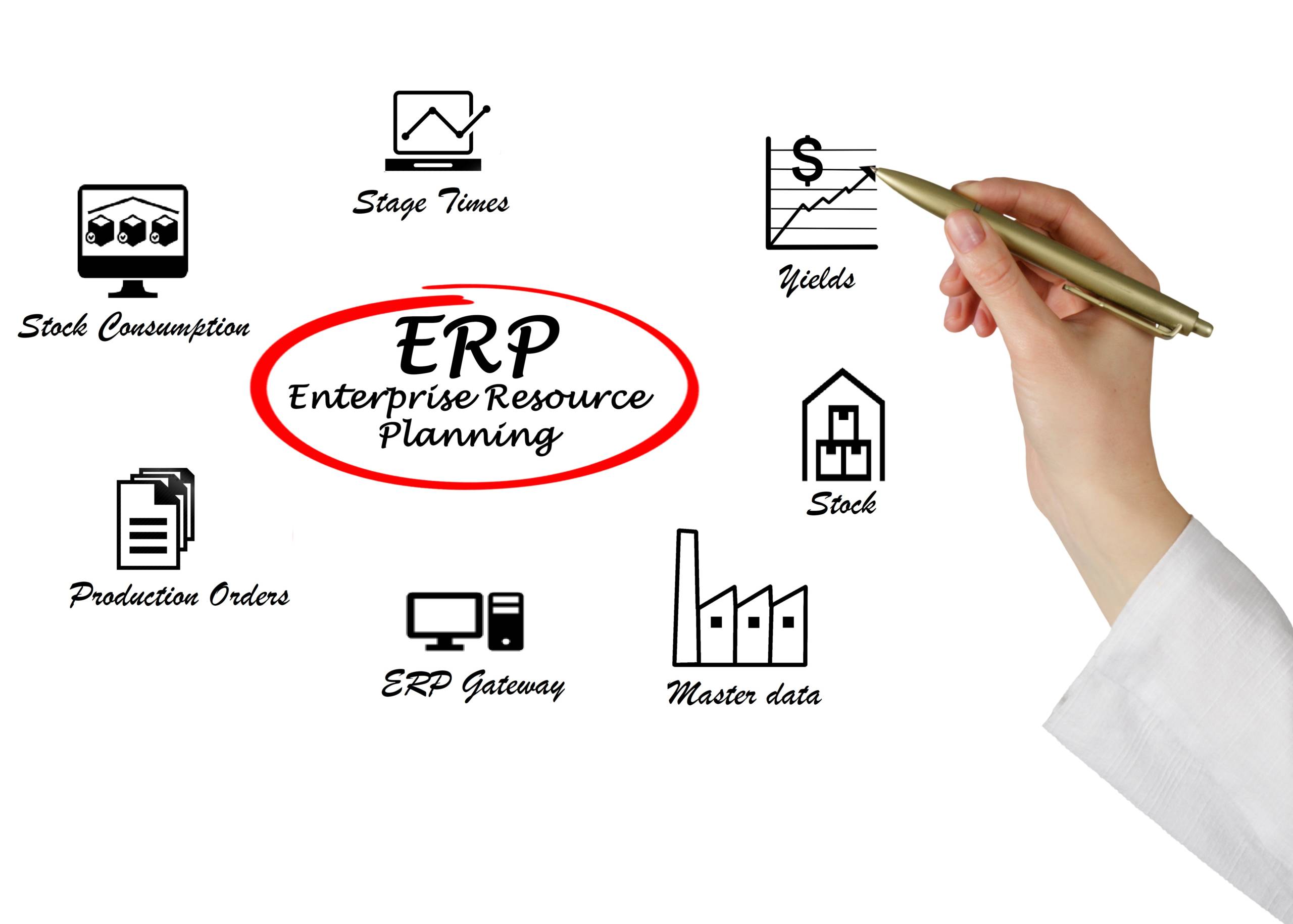
Systems Gateway
The Systems Gateway is a configurable web services interface facilitating communication between the Ci-DMS manufacturing execution system and your ERP system. It has been designed as an open system and is ERP neutral.- Two-way interface for transfer of information such as master data, stock, production orders, stock consumption, stage times, yields
- Proven interfacing with ERP, Warehouse Management and LIMS systems
- Supports data authentication and encryption
- SAP toolbox available for integration with your ECC 6.0 and S/4 HANA environments


Explore More
Continue Your Research
5 Factors to Consider When Choosing Your MES Partner
Selecting the right partner is as important as choosing the right MES solution. We understand..
Contact UsDownload the Ci-DMS MES brochure
Learn more about our Ci-DMS MES — our powerful, versatile, and modular software for paperless manufacturing that helps you tackle even the most exacting and demanding production requirements.
DownloadAutomated, high-precision weight sorting for superior quality control
When Harwell Dosimeters wanted to control the manufacturing quality of their Alanine Pellets they chose CI Precision’s weight sorters to accurately weigh each pellet.
Contact UsInterested in Ci-DMS MES?
To find out more, please contact our team of MES specialists.
Get contact details